Notice
This Item is Out of Stock
This item will ship via Express 1-2 day mail with a flat rate fee of $36.95 and will be packed in an insulated box with ice packs. Items that are required to ship this method will be listed in the item description. Orders placed after 2:00 PM ET (11:00 AM PT) may not end up being able to ship out the same day.
Restrictions: All foreign orders are ineligible. Free shipping is ineligible.
Note: Vaccine orders are only shipped out on Mon-Wed. due to Express service only being able to deliver during weekdays. We cannot be responsible for vaccine or other refrigerated orders that are delayed because no one is present at the time it is delivered.
Description
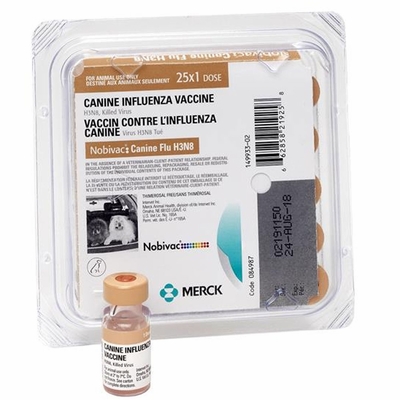
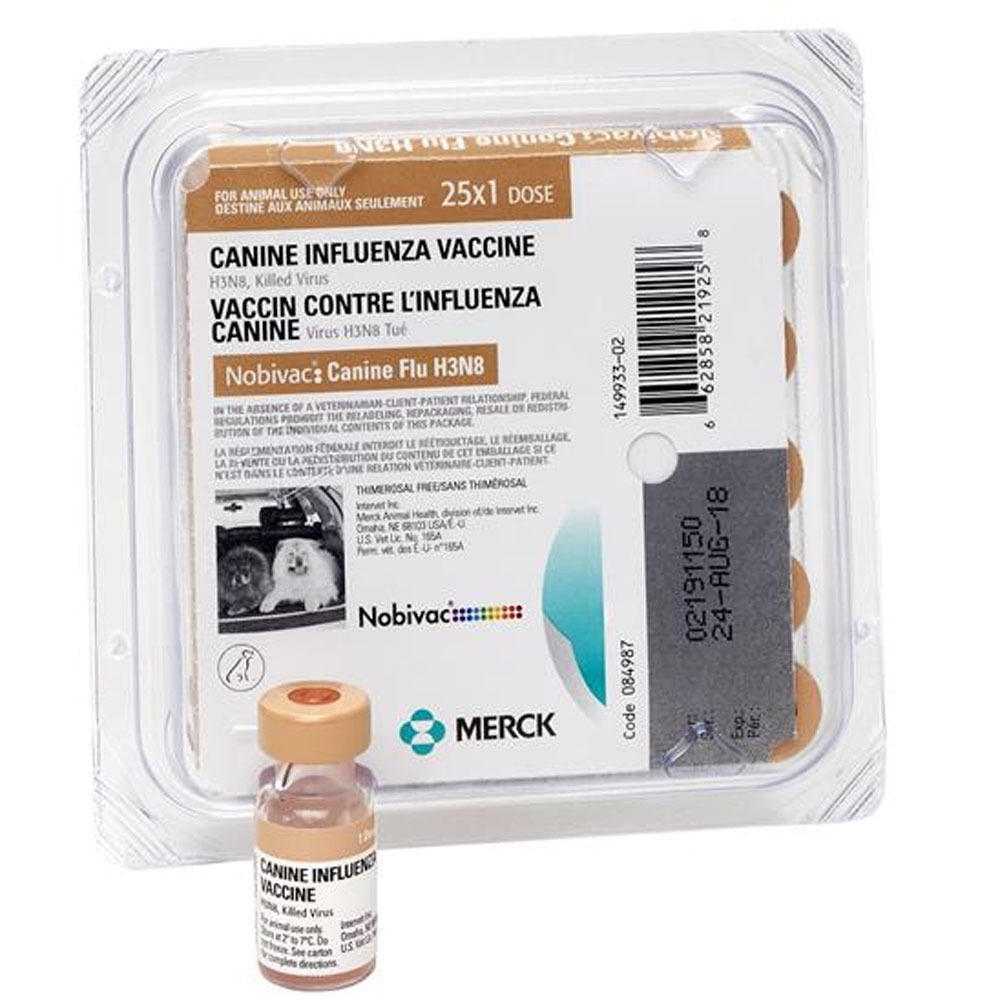
Canine Influenza Vaccine, H3N8 KILLED VIRUS
The first licensed vaccine to protect against canine influenza, recommended for puppies and dogs in the following lifestyle situations:
Features and Benefits
Indications
Nobivac is a registered trademark of Intervet Inc., d/b/a Merck Animal Health, a subsidiary of Merck & Co. Inc.
Useful Information
Safety Information
PRECAUTIONS:
WARNINGS: